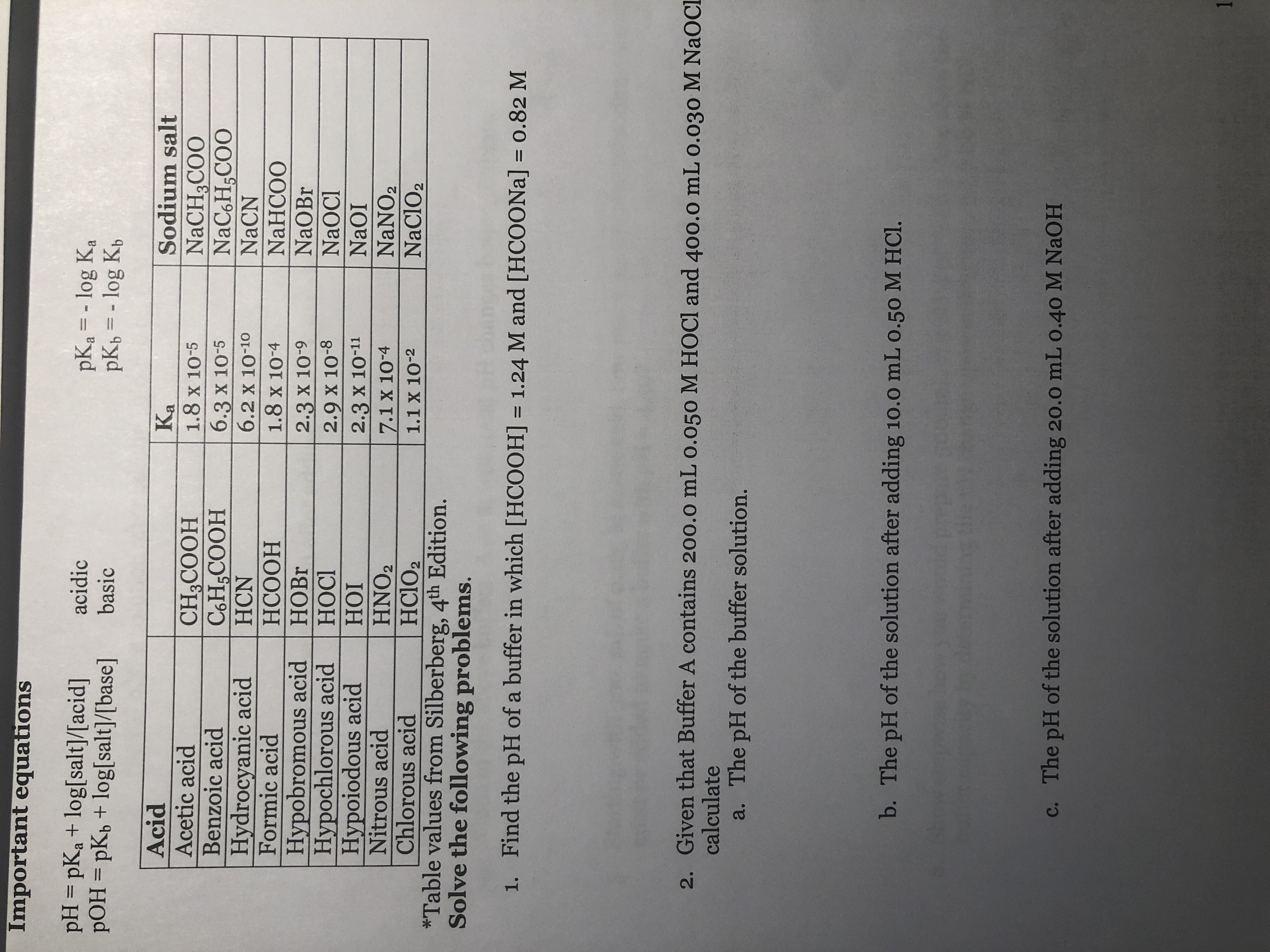
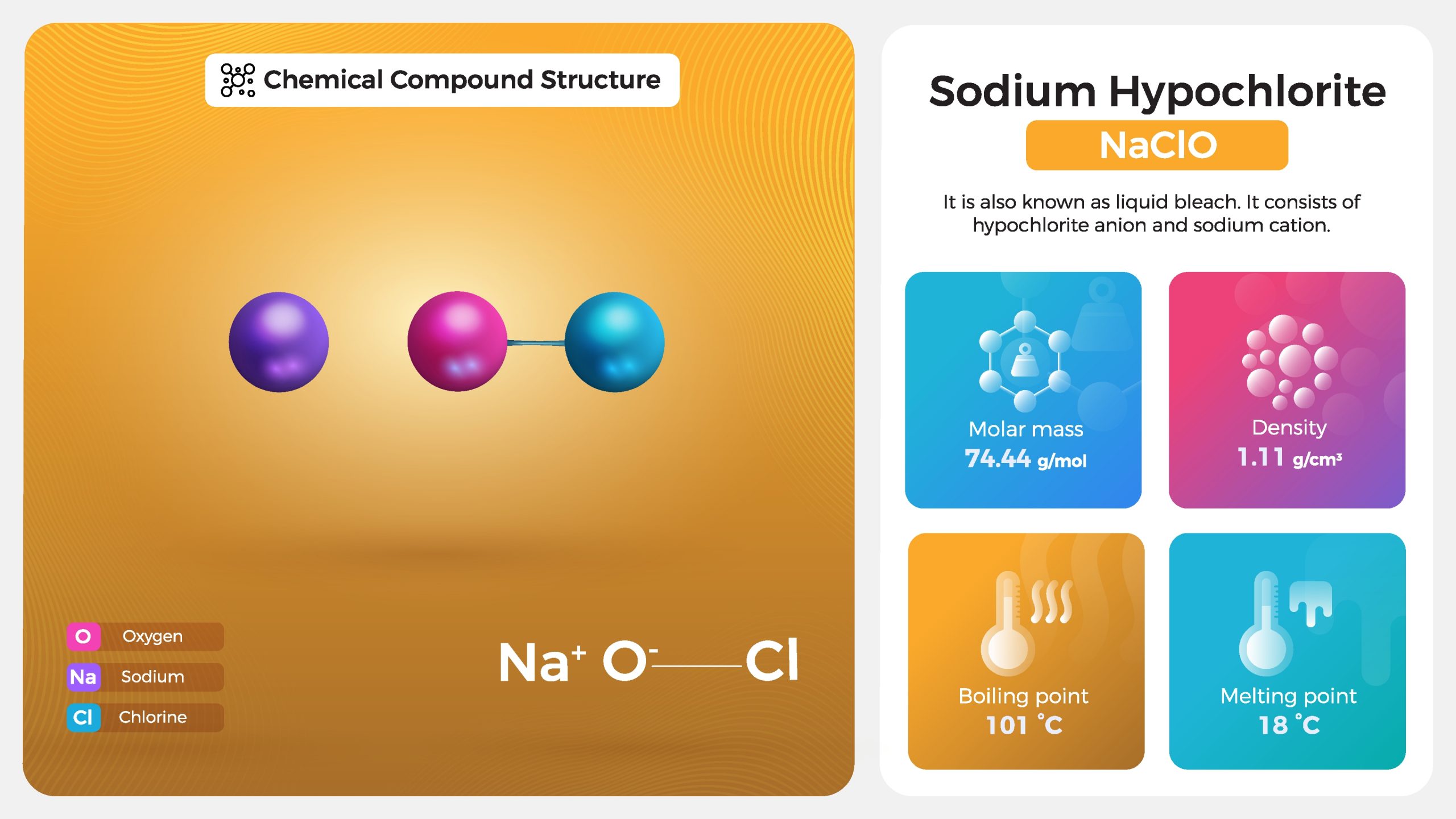
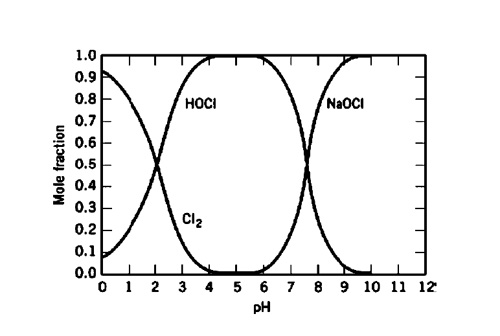
Difference Between Sodium Hypochlorite and Hypochlorous Acid | Compare the Difference Between Similar Terms

SOLVED: Match the following salts with the acid-base neutralization reactants that would produce them: NaCIO4 A HCI + Ca(OH)z NaOCl B. HOCI + Ca(OH)z Ca(OCl)z C.HCI + NaOH D.HCIO4 Ca(OH)2 E: HCIO4 +

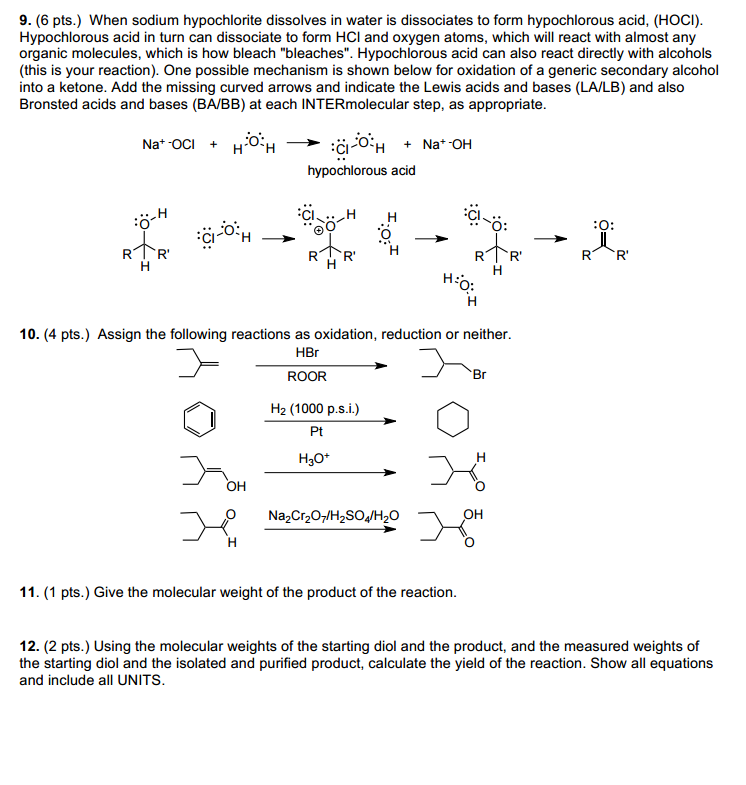
Experiment 19: OXIDATION OF 9-FLUORENOL. Objectives: To synthesize a ketone from a secondary alcohol using household bleach. To purify product using. - ppt download

Difference Between Sodium Hypochlorite and Hypochlorous Acid | Compare the Difference Between Similar Terms